Top-up percutaneous tibial nerve stimulation in MS: an audit
Poster
Introduction
More than 80% of people with MS will have bladder dysfunction over the course of their disease. NICE guidelines CG148 recommend those with overactive bladder (OAB) symptoms refractory to first line treatment of bladder and pelvic floor training and anticholinergic medications, trial sacral nerve modulation. More recently, percutaneous tibial nerve stimulation (PTNS) has been shown to be an effective and safe minimally invasive neuro-modulatory technique to treat refractory overactive bladder symptoms [1][2]. The tibial nerve is a mixed nerve arising from spinal roots L4 to S3, the same spinal segments which innervate the lower urinary tract and the pelvic floor. [3] The exact mechanism of why tibial nerve stimulation improves OAB symptoms is unknown, but is thought to inhibit detrusor activity through spinal inhibitory interneurons via afferent and efferent pathways. [4] In non-neurological patients, up to 71 % had improvement in their OAB symptoms after PTNS.[5]
PTNS has been shown to significantly improve OAB symptoms in up to 89% of MS patients [6][4], and improve all urinary parameters [7]. It is a well-tolerated treatment, and neurological patients tend to be interested in carrying on maintenance or ‘top-up’ maintenance treatments [8]. The improvements that occur at 12 weeks are sustained beyond 12 months.[9][10] This audit will review those who continue with ‘top-up’ PTNS treatment in MS, with a view to identifying characteristics and frequency of treatments.
Methods
This was a retrospective audit evaluating maintenance PTNS treatments in a specialist Uro-Neurology unit at UCLH in pwMS from January 2012 to December 2020. Inclusion criteria were completion of the standard protocol treatment initiation with 12 once-weekly, 30 minute sessions whereby the tibial nerve was stimulated with a 34-gauge needle.[11] pwMS were asked to complete questionnaires and 3-day bladder diaries prior to treatment, and again at the end of 12 weeks of treatment. Changes in OAB symptoms and quality of life were assessed using the validated International Consultation on Incontinence Questionnaires (ICIQ: ICIQ-OAB and ICIQ-LUTSqol, with a maximum score of 16 and 76, respectively). A satisfaction survey was completed at the end of treatment and request to continue with additional treatments after completion of 12 weeks and patients who had a significant benefit and requested to continue top-ups were invited to continue in the maintenance phase whereby they attended ‘top-up’ treatments when self-reporting a recurrence of their symptoms. A maintenance treatment consisted of a single session of 30 minutes stimulation. Patients were identified from our PTNS database, and electronic case notes were scrutinised for the following detail: demographics, date of PTNS treatments, subsequent intra-detrusor botulinum toxin treatment and duration of MS. The top-up treatments were categorised into time epoch from the date of PTNS initial treatment completion i.e. less than 3 months, 3 to 6 months, 6 to 12 months, 12 to 18 months, 18 months to 2 years, 2 to 3 years, 3 to 4 years and 4 years plus.
Results
A total of 43 MS patients (34, 77% female ) completed an initial course of PTNS (Table 1). 26 patients (59%) requested on-going top-up PTNS treatment. The mean age of the top-up group was 45.6 years (95% CI 40.4, 51.1) compared with the no top-up group 48.6 years (95% CI 43.1, 50.1). The duration of MS was similar for the two groups (Table 1).
Overall for the entire cohort the recorded ICIQ-OAB and ICIQ-LUTSqol, and urinary frequencies were similar after 12 weeks of initial treatment. Both groups experienced a reduction in mean daily urinary frequency after treatment.
The median number of PTNS top-ups per patient was 8 (range 1-53), with the mean top-up treatment occurring every 3.5 months (Figure 1). The median duration of returning for top-up treatments was 26 months. 35% of all top-up treatments occurred within the first 12 months after completing the initial treatment (Figure 2). Nearly two thirds (65%) of patients continued top-ups for at least 24 months.
Covid-19 pandemic has caused significant disruption to the delivery of PTNS top-ups, with only four patients in the top-up group receiving treatment after March 2020. One patient subsequently purchased a transcutaneous tibial nerve stimulation machine. One patient from the top-up group started intra-detrusor botulinum toxin injections.
Of the no top-up group, 3 patients started intra-detrusor botulinum toxin injections, and one patient is awaiting transcutaneous tibial nerve stimulation.
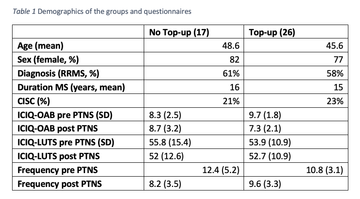
Table 1 Demographics of the groups and questionnaires
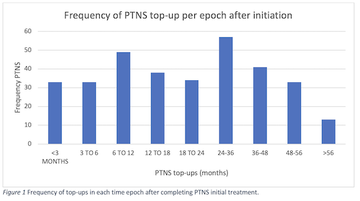
Figure 1 Frequency of top-ups in each time epoch after completing PTNS initial treatment.
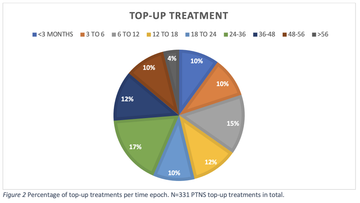
Figure 2 Percentage of top-up treatments per time epoch. N=331 PTNS top-up treatments in total.
Discussion
Lower urinary tract symptoms are common amongst MS patients, and pwMS often have symptoms refractory to first line treatments, or the side effects of anti-cholinergic medications are not acceptable. Phase 3 clinical trials of PTNS to manage OAB symptoms have excluded neurological patients, but it does appear to be efficacious.[8] The mechanism of PTNS is not well understood, but studies have demonstrated reduction in incontinence episodes, voiding frequency, and nocturia in MS. [4,10][9]
In our cohort, the top-up group who continued treatment, and the no top-up group had similar outcomes at the end of their initial 12 week trial. There were no differences within the cohorts and their quantitative response to initial 12 weeks PTNS treatment. Both groups had improvement in urinary frequency. Their subjective satisfaction on improvement of symptoms may have been different and contributed to continuing with top-ups. This was not analysed in this audit. The sample size was small and lacked statistical power to make statistical inference. The long-term improvement in OAB symptoms in the top-up group is not quantified but given two thirds of patients continued top-ups for up to 2 years, there was at least subjective improvements in OAB symptoms.
PTNS appears to be a promising second-line treatment of OAB symptoms in pwMS and is well-tolerated. A significant barrier for undergoing PTNS treatment is the travel and time commitments (weekly for 12 weeks). Availability of transcutaneous technology will hopefully reduce this barrier, and also ensure ongoing treatment for example during the Covid-19 pandemic.
In conclusion, there is a paucity of longitudinal PTNS data in pwMS, which requires further research. Key to our understanding will be – what is the long-term outcome of PTNS top-up on OAB symptoms in MS, and how to increase the accessibility to treatment.
References
- Peters KM, Carrico DJ, Perez-Marrero RA, et al. Randomized trial of percutaneous tibial nerve stimulation versus Sham efficacy in the treatment of overactive bladder syndrome: results from the SUmiT trial. J Urol 2010;183:1438–43. doi:10.1016/j.juro.2009.12.036
- Percutaneous posterior tibial nerve stimulation for overactive bladder syndrome. 2020;:1–6.
- McGuire EJ, Zhang SC, Horwinski ER, et al. Treatment of motor and sensory detrusor instability by electrical stimulation. J Urol 1983;129:78–9. doi:10.1016/s0022-5347(17)51928-x
- De Sez M, Raibaut P, Gallien P, et al. Transcutaneous Posterior Tibial Nerve Stimulation for Treatment of the Overactive Bladder Syndrome in Multiple Sclerosis : Results of a Multicenter Prospective Study. 2011;311:306–11. doi:10.1002/nau
- Enrico F-A, Filomena P, Francesco S, et al. Percutaneous Tibial Nerve Stimulation Effects on Detrusor Overactivity Incontinence are Not Due to a Placebo Effect: A Randomized, Double-Blind, Placebo Controlled Trial. J Urol 2010;184:2001–6. doi:10.1016/j.juro.2010.06.113
- Gobbi C, Digesu GA, Khullar V, et al. Percutaneous posterior tibial nerve stimulation as an effective treatment of refractory lower urinary tract symptoms in patients with multiple sclerosis: preliminary data from a multicentre, prospective, open label trial. doi:10.1177/1352458511414040
- Kabay S, Kabay C, Yucel M, et al. The Clinical and Urodynamic Results of a 3-Month Percutaneous Posterior Tibial Nerve Stimulation Treatment in Patients With Multiple Sclerosis-Related Neurogenic Bladder Dysfunction. 2009;968:964–8. doi:10.1002/nau
- Tudor KI, Seth JH, Liechti MD, et al. Outcomes following percutaneous tibial nerve stimulation (PTNS) treatment for neurogenic and idiopathic overactive bladder. Clin Auton Res 2020;30:61–7. doi:10.1007/s10286-018-0553-8
- Zecca C, Digesu GA, Robshaw P, et al. Maintenance percutaneous posterior nerve stimulation for refractory lower urinary tract symptoms in patients with multiple sclerosis: an open label, multicenter, prospective study. J Urol 2014;191:697–702. doi:10.1016/j.juro.2013.09.036
- Kabay SC, Kabay S, Mestan E, et al. Long Term Sustained Therapeutic Effects of Percutaneous Posterior Tibial Nerve Stimulation Treatment of Neurogenic Overactive Bladder in Multiple Sclerosis Patients : 12-Months Results. 2017;110:104–10. doi:10.1002/nau
- Peters KM, Carrico DJ, Wooldridge LS, et al. Percutaneous Tibial Nerve Stimulation for the Long-Term Treatment of Overactive Bladder : 3-Year Results of the STEP Study. JURO 2013;189:2194–201. doi:10.1016/j.juro.2012.11.175
More MS Academy Bladder and bowel Projects
Encouraging excellence, developing leaders, inspiring change
MS Academy was established in 2016 and in that time has accomplished a huge amount with exciting feedback demonstrating delegates feel inspired and energised along their personal and service development journeys. The various different levels of specialist MS training we offer are dedicated to case-based learning and practical application of cutting edge research.